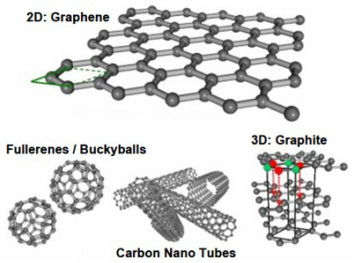
Different formations of carbon atoms [4]
Structure
It is one single layer of crystalline graphite, or carbon atoms, which are bonded together to form hexagonal shapes and arranged in a chicken wire or honeycomb formation. It is a 2-dimensional material that measures only 1 atom in thickness. [1]
Properties
Layers of graphene contain very strong intermolecular forces between the Carbon atoms, allowing it to be a very strong material and highly heat resistant. Graphene structures can survive heats of up to 300 ºC in air and even higher temperatures when placed in a vacuum, making it an excellent thermal conductor. The strength of the bonds allows the material to withstand vibrations of high frequencies without breaking the bonds. [3]
Some unique properties of graphene include its flexibility, stretchability and excellent ability to conduct electricity with almost no resistance on the flow of electrons. This conductive property holds true at all temperatures, not exclusively at high or low ones, and would make this material very useful in creating computer circuits and electronic microchips with no resistance. [3] Another unique property of graphene is that it is nearly transparent. When light is shined on it, it only absorbs 2.3% of the light. This property makes it a great option for use in manufacturing touchscreens.[3]
It is one single layer of crystalline graphite, or carbon atoms, which are bonded together to form hexagonal shapes and arranged in a chicken wire or honeycomb formation. It is a 2-dimensional material that measures only 1 atom in thickness. [1]
Properties
Layers of graphene contain very strong intermolecular forces between the Carbon atoms, allowing it to be a very strong material and highly heat resistant. Graphene structures can survive heats of up to 300 ºC in air and even higher temperatures when placed in a vacuum, making it an excellent thermal conductor. The strength of the bonds allows the material to withstand vibrations of high frequencies without breaking the bonds. [3]
Some unique properties of graphene include its flexibility, stretchability and excellent ability to conduct electricity with almost no resistance on the flow of electrons. This conductive property holds true at all temperatures, not exclusively at high or low ones, and would make this material very useful in creating computer circuits and electronic microchips with no resistance. [3] Another unique property of graphene is that it is nearly transparent. When light is shined on it, it only absorbs 2.3% of the light. This property makes it a great option for use in manufacturing touchscreens.[3]
Works Cited
1 Brooks, Michael. "It's Super-Carbon!." New Scientist 204.2736 (2009): 48. Science Reference Center. Web. 27 Nov. 2012.
2 Duffin, Angeli Duffin. Graphene Structure. N.d. Photograph. Earth Techling. 27 Jan. 2012. Web. 9 Dec. 2012. <http://c276521.r21.cf1.rackcdn.com/wp-
content/uploads/2012/01/Graphene.jpg>.
3 Geim, Andre, and Antonio Castro Neto. "Graphene." New Scientist 214.2863 (2012): i. Science Reference Center. Web. 7 Dec. 2012.
4 Kraft, Rudolf. Different Structures of Graphenes. 6 January 2011. 6 December 2012. <http://evworld.com/article.cfm?storyid=1950>.
2 Duffin, Angeli Duffin. Graphene Structure. N.d. Photograph. Earth Techling. 27 Jan. 2012. Web. 9 Dec. 2012. <http://c276521.r21.cf1.rackcdn.com/wp-
content/uploads/2012/01/Graphene.jpg>.
3 Geim, Andre, and Antonio Castro Neto. "Graphene." New Scientist 214.2863 (2012): i. Science Reference Center. Web. 7 Dec. 2012.
4 Kraft, Rudolf. Different Structures of Graphenes. 6 January 2011. 6 December 2012. <http://evworld.com/article.cfm?storyid=1950>.
Word count: 196
